Introduction
Because energy production is a major source of greenhouse gas emissions, engineers and scientists are exploring alternative energy systems that reduce carbon emissions. One option being studied is hydrogen as an energy carrier, which can be produced in different ways with very different environmental impacts.
Part 1: Hydrogen Fuel
Instructions: Read the text below to learn more about hydrogen production, and then answer the comprehension questions that follow.
Hydrogen gas can be used as an energy carrier, meaning it stores energy that can later be converted into electricity or heat. Hydrogen is often described using color labels that refer to how it is produced, not the hydrogen itself.
Hydrogen Color Labels
Hydrogen is the most abundant element in the universe, but it is rarely found on Earth as a pure gas. Instead, hydrogen is usually bonded to other elements, such as oxygen in water (H2O) or carbon in natural gas or coal. Energy is required to separate hydrogen from these compounds.
Hydrogen itself does not contain carbon, but the process used to produce hydrogen can result in different levels of carbon dioxide emissions. To describe these differences, scientists, engineers, and policymakers often use color labels. These labels do not describe the hydrogen’s physical properties; instead, they describe how the hydrogen is produced.
Gray hydrogen is produced from hydrocarbons, most commonly natural gas which is primarily composed of methane (CH₄), using a process called steam methane reforming (SMR). In this process, methane reacts with steam at high temperatures to produce hydrogen gas and carbon dioxide.
- Hydrogen is the desired product.
- Carbon dioxide is released into the atmosphere.
- Gray hydrogen is currently the most common form of hydrogen production worldwide.
Because CO2 is released during production, gray hydrogen is associated with significant greenhouse gas emissions.
Blue hydrogen is produced using the same basic process as gray hydrogen (steam methane reforming), but with an added step: carbon capture and storage (CCS).
- Carbon dioxide produced during hydrogen generation is captured.
- The captured CO2 is stored underground or used in other industrial processes.
- Some emissions may still occur due to incomplete capture or energy use.
Blue hydrogen generally results in lower carbon dioxide emissions than gray hydrogen, but it does not eliminate emissions entirely.
Black or brown hydrogen is produced from coal using a process called coal gasification. In this process, coal reacts with steam and oxygen at high temperatures to produce hydrogen gas and carbon dioxide.
- Hydrogen is the desired product.
- Carbon dioxide is released into the atmosphere.
- Used in some regions where coal is widely available.
Because coal contains more carbon than natural gas, black or brown hydrogen is associated with higher greenhouse gas emissions than gray hydrogen.
Green hydrogen is produced using electrolysis, a process that uses energy to split water (H2O) into hydrogen (H2) and oxygen (O2).
- No carbon-containing compounds are used in the reaction.
- The electrolysis process itself does not produce carbon dioxide.
- The overall environmental impact depends on how the electricity is generated.
If the electricity used for electrolysis comes from low-emission sources such as wind, solar, or hydropower, the hydrogen is considered green hydrogen because no direct carbon emissions occur during production.
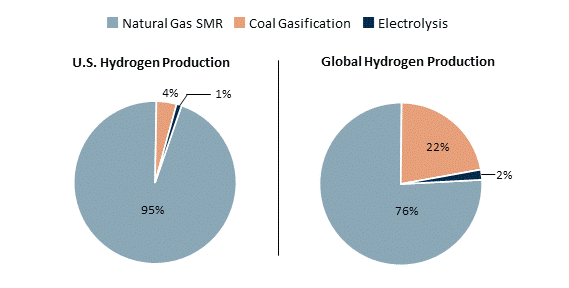
The graph below shows how hydrogen is currently produced in the U.S. and around the world. It highlights the dominant role of hydrocarbon-based hydrogen, as well as the relatively small share produced using electrolysis. Although electrolysis can produce hydrogen with very low emissions, it currently represents a small share of hydrogen production because of its high cost and need for large amounts of electricity.
Comprehension Questions
- What is meant by “gray” hydrogen and “blue” hydrogen? How are they produced?
- Why do many current hydrogen production methods result in carbon dioxide emissions?
- How does electrolysis work, and what role does electricity play in the process?
- What is “green” hydrogen and how does it differ from other types of hydrogen?
- What does the graph show about how hydrogen is produced in the U.S. and globally?
- Why does electrolysis currently only make up a tiny share of hydrogen production?
Part 2: Electrolysis
Read the text below to learn more about electrolysis, and then answer the comprehension questions that follow.
Electrolysis is a chemical process that uses electrical energy to drive a non-spontaneous chemical reaction, meaning a reaction that would not occur on its own. This process takes place in an electrolytic cell, which contains two electrodes—an anode and a cathode—immersed in an electrolyte, a substance that allows ions to move freely.
In an electrolytic cell, the anode becomes positively charged, and the cathode becomes negatively charged. When an external power source supplies electricity to the system, electrons are forced to move through the circuit, causing chemical reactions to occur at each electrode.
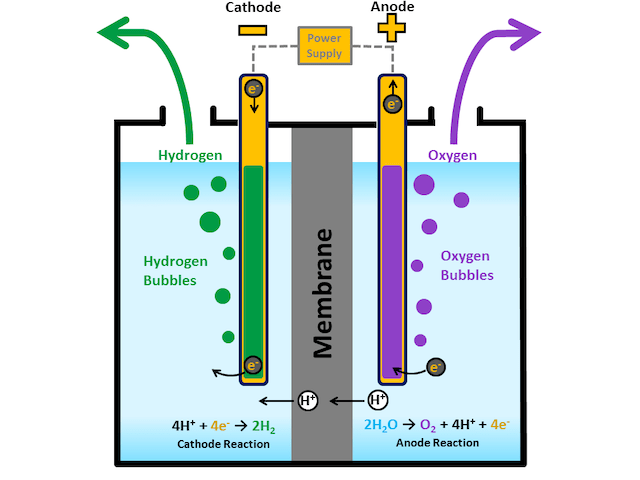
During electrolysis, oxidation occurs at the anode, while reduction occurs at the cathode.
A common way to remember this is LEO goes GER.
- Loss of Electrons is Oxidation.
- Gain of Electrons is Reduction.
Oxidation means a substance loses electrons, while reduction means a substance gains electrons. The substances that undergo oxidation or reduction may be ions dissolved in the electrolyte or atoms that are part of the electrode material itself.
A common example of electrolysis is the electrolysis of water. In this process, water molecules (H₂O) are broken down into hydrogen gas (H₂) and oxygen gas (O₂).
Looking at the half-reactions helps clearly show where electrons are gained and lost.
- At the cathode (reduction), hydrogen ions (H⁺) gain electrons and form hydrogen gas:
2H++2e− → H2
- At the anode (oxidation), oxygen ions (O²⁻) lose electrons and form oxygen gas:
2H2O → O2+4H++4e−
- When the half-reactions are combined, the overall reaction can be summarized as:
2H₂O → 2H₂ + O₂
The electrodes used in electrolysis are typically made from inert metals, such as platinum or iridium. These metals are chemically stable and do not react with the electrolyte or the products of the reaction. Platinum is commonly used because it is an excellent conductor of electricity and remains stable in both water and oxidizing environments.
The amount of substance produced during electrolysis depends on how much electric charge moves through the system. This charge is determined by how strong the electric current is and how long it flows. In general, the more electricity that passes through the cell, the more hydrogen gas is produced.
Electrolysis has many important applications in science and industry. It is used to produce hydrogen gas, purify and extract metals, and coat objects through electroplating. Electrolysis is also essential in industrial processes such as the production of chlorine gas and sodium hydroxide, both of which are widely used in manufacturing and chemical industries.
Comprehension Questions
- Explain oxidation and reduction in the context of electrolysis. Which process begins at the anode, and which occurs at the cathode?
- Describe the electrolysis of water. Include the products formed, and the reactions that take place at each electrode.
- What are some practical applications of electrolysis in industry or technology?
Part 3: Building an Electrolytic Cell
In this activity, you will construct a simple electrolytic cell and use electrical energy to split water into hydrogen and oxygen gas. You will observe gas formation at each electrode and measure the amount of hydrogen produced.
Important Safety Information
This lab uses low-voltage electricity and produces small amounts of hydrogen gas. Hydrogen is flammable, so no flames or heat sources are allowed. Wear safety goggles, handle sharp and hot tools carefully, and disconnect the battery when finished.
Materials
- Beaker
- 2 clear plastic straws
- Optional: Pipette dropper for filling straw with water
- 2 graphite pencils (sharpened at both ends)
- 2 jumper wires with alligator clips
- 9V battery
- Masking tape
- Scissors
- Hot glue (or another method of sealing the straw)
- Waterproof marker
- Tap water
- Ruler
- Safety goggles
Procedure
- Sharpen both ends of each pencil so that each end is identical and graphite is visible on both ends.

2. Seal one end of one of the drinking straws completely so that no water or gas can escape. To do this, fold one end and hot glue it closed to get a good seal.

3. Cut a small hole toward the open end of the same straw so that the entire graphite tip of the pencil can fit snugly inside the drinking straw.
Note: In the picture, the pencil is shown for reference.

- Fill the beaker about halfway with tap water.
- Fill the sealed straw completely with tap water.


- To make sure no air bubbles remain, cut the second straw in half to create a long, thin piece. Holding the hole you cut in the straw so that no water leaks, use this piece to get rid of air bubbles in the sealed straw, as shown in the second picture above.
- Place the straw vertically into the beaker, with the sealed end facing up, without spilling any water. The straw must remain completely full. Tape the straw to the side of the beaker so it does not move.
9. Tape the two pencils together and place them into the water. Make sure one pencil (the cathode) is positioned with its graphite tip inside the straw.
Note: It’s important to make sure the whole graphite section of the pencil is snug inside the straw, or else gas bubbles will escape the straw.

- Clip the jumper wires to the graphite at the dry end of each pencil (not in the water) and connect the graphite to the 9V battery using the jumper wires. Using the marker, label the pencil connected to the negative terminal as the cathode (-). Label the pencil connected to the positive terminal as the anode (+).
Note: Remember that the cathode should be the pencil positioned inside the straw. Connect it to the negative terminal.
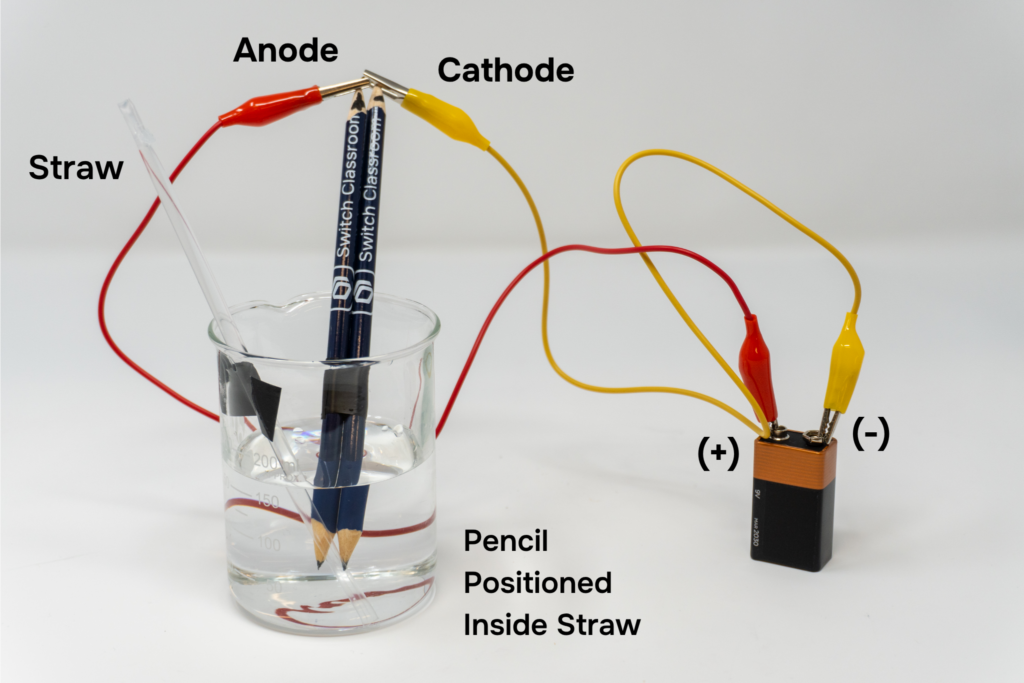
- Using the waterproof marker, mark the initial water height on the side of the straw and record the measurement in the data table.
- Allow the electrolytic cell to run for 10 minutes. Observe gas formation at both electrodes.
- After the reaction time has passed, mark the final water height on the side of the straw and record the measurement in the data table.
Observation
Insert or sketch a picture of your electrolytic setup here. Add labels.
Data Table 1: Electrolysis Measurements
| Measurement | Value and Unit |
|---|---|
Initial Water Height | |
Final Water Height | |
Change in Water Height (Final – Initial) |
Analysis Questions
- Did gas collect inside the straw as expected? If yes, describe the volume of gas collected based on observation. If not, what might have gone wrong in your setup?
- Which gas was supposed to collect inside the straw?
- At which electrode (anode or cathode) was this gas produced? Explain how you know based on the electrolysis of water.
- What factors in your experimental setup may have limited the amount of hydrogen produced?
- What improvements or changes would you make to this experiment if you were to do it again, and what would be the predicted results of those changes?
- How might industrial electrolyzers be designed to increase hydrogen production while minimizing energy use and emissions?
- If the electricity used for this experiment came from low-carbon electricity sources, such as solar or wind, what would be the color classification of the hydrogen produced?
