Bell Ringer
Instructions: Select one of the Bell Ringers for students to reflect on and answer.
Vocabulary
Instructions: Go over important terms and their definitions before watching the Introduction to Nuclear video. Student vocabulary list can be found in the Student Guide and Introduction to Nuclear – Starter Pack.
| Word | Definition | Example |
|---|---|---|
| Proliferation | noun; the rapid spread or reproduction of something | “Another challenge is proliferation. Could terrorists turn this waste into a bomb?” |
| Radioactive | adjective; radioactive elements give off particles and energy that can be harmful if not handled safely | “It still has a huge amount of energy in it, which means it’s hot, and it’s radioactive.” |
| Spent fuel | noun phrase; used up nuclear fuel that can’t be used, but is still very hot and dangerous | “Handling this spent fuel is a major challenge of nuclear . . .” |
| Cooling tower | noun phrase; a tall structure where water is cooled and evaporated by exposing it to air | “That’s steam coming out of the cooling tower; water is evaporated to cool the plant.” |
| Emissions | noun; solid, liquid or gas particles that are released from an object or when a substance is burned | “Nuclear [makes electricity] with zero emissions.” |
| Pellet | noun; a small, solid piece of something – like a tiny fuel capsule | “One uranium pellet weighs a quarter of an ounce.” |
| Equivalent | adjective; the same amount or value as something else | “That’s equivalent to one ton of coal.” |
| Robust | adjective; very strong and able to hold up under tough conditions | “To contain all that energy, the plants must be incredibly robust.” |
| Opposition | noun; people who disagree with something and try to stop it | “That means public opposition, lawsuits, more time, and more expense to build a plant.” |
| Plant | noun; a large building where electricity or other products are made | “They’re much more expensive than, say, a natural gas plant.” |
| Uranium | noun; a naturally radioactive metal element used as fuel in nuclear energy | “One uranium pellet weighs a quarter of an ounce.” |
Quiz & Cloze Notes
Instructions: Review key concepts after watching the Introduction to Nuclear video. The Student Guide and Introduction to Nuclear – Starter Pack contain the quiz and cloze notes.
Quiz Answer Key: Q1:C Q2:A Q3:C Q4:C
Cloze Notes Answer Key: nuclear, electricity, powerful, kilowatt, hazards, safety record, challenge, cost
Data Set
Instructions: Provide students with the Introduction to Nuclear – Data Set for data literacy and analysis practice.
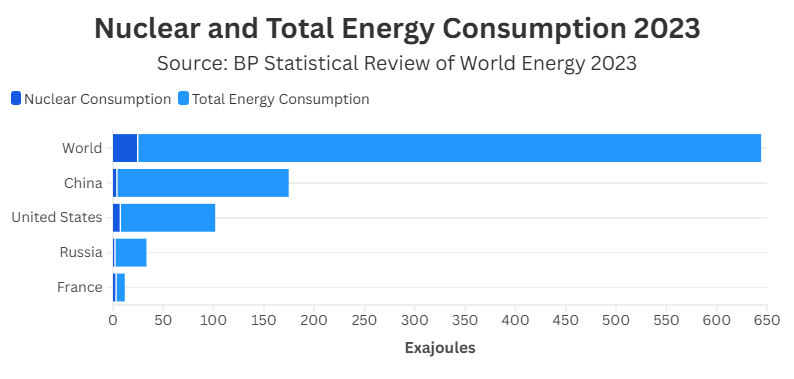
Answer Key: Question 1: France Question 2: United States, France, China, Russia
Question 3: No; The new percentage would be 15%, which is still less than France’s 35%.
Question 4: Answers will vary. (Example: Nuclear energy is a minor component of the global energy system, as it only makes up 4% of the world’s total energy consumption. Question 5: Answers will vary.
Nuclear Reaction in a Box – Hands-On
Introduction
In this hands-on activity, students will model the process of nuclear fission using beads to represent atoms and neutrons. By simulating collisions and tracking the resulting chain reaction, students will see how a single split atom can rapidly multiply to release large amounts of energy. This engaging demonstration builds a concrete understanding of particle motion, energy release, and the concept of chain reactions in nuclear energy.
Student Objectives
Students will be able to
- Model the process of nuclear fission and explain how one split atom can trigger a chain reaction.
- Collect and record data to observe how the number of particles changes over time during a fission simulation.
- Compare how the speed of particle movement affects the rate of energy production in a chain reaction.
Materials
- Student Handout
- 1 container (i.e., shoe box, clear plastic container, etc.) per group
- 1 red bead per group
- 100 green beads (color choices are optional) per group
- Timer
- Groups must have 3+ students who will be the shaker, the timer, and the multiplier(s)
- Optional: Graph paper or digital graphing tool
Procedure
1. Demonstration Setup:
Put one red bead and one green bead in a clear container, with a handful of green beads nearby.

2. Introduction (I do /We do):
Say: Today, we are going to learn about nuclear energy! It’s a special type of energy stored inside tiny building blocks of matter called atoms. We get nuclear energy from an element called uranium. The process we’re learning about today is called nuclear fission. Inside a uranium atom, there is a lot of energy holding it together. When we hit the uranium atom with a tiny particle called a neutron, the atom splits apart into smaller pieces. When this happens, a huge amount of energy is released! This energy can be used to make electricity.
Model the following discussion with students as you demonstrate nuclear fission in the box.
Question: What do you think happens to the energy of one when it is released? (Model one red bead colliding with a green bead. Once they hit, shout “Collision!” and stop moving the container)
Answer: When the uranium atom is split, it breaks into smaller atoms, 2-3 neutrons and LOTS of energy. (Model by adding two green beads. Keep the container still.)
Question: But the atoms don’t stay still like we’ve got them here! They’re full of energy and they’re moving! What will happen if I move the container?
Answer: They’ll collide!
Question: And then what will happen when they collide?
Answer: It will split into smaller pieces and three neutrons will be released. (Model, move the container until two green beads collide. Shout, “Collision!”, add three beads for each collision. Keep the container still.
Question: But the atoms don’t stay still, so what’s going to happen?
Answer: They’ll keep bumping into each other; they’ll keep splitting! We’ll keep adding more beads. (Give two students the bag of green beads and instruct them to shout, “Collision!” and take turns adding three beads each time the beads collide.)

3. Activity Phase 1 (You Do – Slowly):
Tell students that they will do this process twice. The first time, they are to do it slowly while they record their work.
Pass out the worksheet and materials to each group. Have students read the instructions carefully and designate “Timer” “Shaker” and “Multiplier” roles. Following instructions on the student handout, students will empty the container so only one red “starter atom” bead and one green bead are in it. Remind them that they are modeling fission, when one uranium atom collides with a neutron and creates more particles that will continue to collide. When the Timer starts timing, the Shaker can slowly begin to shake the container. Once the “starter atom” red bead hits the green bead, the Multiplier will add three more green beads. The Timer will call out “10 seconds” and the Shaker and Multiplier must pause to observe, record, and make predictions on the worksheet. Fission can resume, beads are added, and the timer will watch for another 10 seconds to pass. When the Timer calls out “20 seconds,” the group will pause to observe and record. The Timer, Shaker and Multiplier will repeat one more time, allowing another 10 seconds to pass before calling out “30 seconds”. The group will pause to observe and record. Using the attached worksheet, students will record the following:
- After shaking the box for 10 seconds, count how many green beads you have; this represents the amount of energy you have.
- Predict what will happen if you do this for another 20 seconds. 30 seconds? A minute? An hour? A day?
- Test this by shaking the box for 20 seconds, and then 30 seconds.
4. Activity Phase 2 (You Do – Fast):
Continuing with the simulation, students will get ready for the second round. Students will repeat the above steps except this time, the Shaker can move the container quickly to see if the Multiplier(s) can keep up.
Students will clear the container out and go back to one red and one green bead, answering the following questions.
- What happens if you shake the box harder? (Should generate energy more quickly).
- Now, test the speed of the box by shaking faster. (Repeat steps above at the faster speed).
Optional Extension:
Make graphs for both trials of Actual Quantity v. Time.
Answer Key
The Student Guide contains the Nuclear Reaction in a Box – Student questions.
Student Questions Sample Responses
A. Fission is when a uranium atom is hit by a neutron and splits. This collision releases energy and produces more neutrons that can hit other uranium atoms, causing a chain reaction of splitting atoms.
B. The red “starter atom” would hit green beads more quickly, so more green beads would be added in a shorter time. The chain reaction would happen faster.
C. There would be a huge number of green beads and the box might overflow because the reaction keeps multiplying very quickly.
D. The green beads represent neutrons released during fission that can cause more uranium atoms to split.
E. It would cause an extremely fast chain reaction, releasing massive amounts of energy all at once – this could cause a nuclear explosion.
Exit Ticket
Instructions: Access the Exit Ticket and have students reflect on and answer the prompt.
