Bell Ringer
Instructions: Select one of the Bell Ringers for students to reflect on and answer.
Vocabulary
Instructions: Go over important terms and their definitions before watching the Introduction to Natural Gas video. Student vocabulary list can be found in the Student Guide and Introduction to Natural Gas – Starter Pack.
| Word | Definition | Example |
|---|---|---|
| Versatile | adjective: having many uses or applications | “Natural gas is versatile, abundant and cleaner.” |
| Abundant | adjective: present in great quantities (as of resources) | “Natural gas is versatile, abundant and cleaner. |
| Consumers | noun: one that utilizes economic goods | “And in the US, and many other countries, including China and India, the big energy consumers of the developing world, hydraulic fracturing is producing natural gas where it was previously not possible.” |
| Developing World | noun: a portion of the emerging and developing world that has a low economic level of industrial production and standard of living because of lack of capital | “And in the US and many other countries, including China and India, the big energy consumers of the developing world, hydraulic fracturing is producing natural gas where it was previously not possible.” |
| Hydraulic Fracturing | noun: a drilling completion process that brings saltwater, natural gas and oil to the surface. If the oil well is not handled properly near the surface, it can sometimes leak into local groundwater | “And in the US and many other countries, including China and India, the big energy consumers of the developing world, hydraulic fracturing is producing natural gas where it was previously not possible.” |
| Greenhouse Gas | noun: a gas that contributes to the greenhouse effect by absorbing infrared radiation, e.g., carbon dioxide, methane and chlorofluorocarbons | “Natural gas itself is also a greenhouse gas, 20 times more potent than CO2, so we’ll need to minimize accidental releases of it. |
| Fracking | noun: shorthand for hydraulic fracturing | “This has got some people worried that fracking will contaminate water supplies and government regulators are looking hard at this issue.” |
| Carbon Emissions | noun: refers to CO2. An odorless, colorless gas that is discharged into the atmosphere from the burning of fossil fuels and the manufacturing of cement | “So natural gas is a cheap, abundant power source that could help all of us reduce our carbon emissions.” |
Quiz & Cloze Notes
Instructions: Review key concepts after watching the Introduction to Natural Gas video. The Student Guide and Introduction to Natural Gas – Starter Pack contain the quiz and cloze notes.
Quiz Answer Key: Q1:C Q2:A Q3:D Q4:B
Cloze Notes Answer Key: carbon, hydraulic, fracturing, fossil, carbon dioxide, greenhouse gas
Data Set
Instructions: Provide students with the Introduction to Natural Gas – Data Set for data literacy and analysis practice.
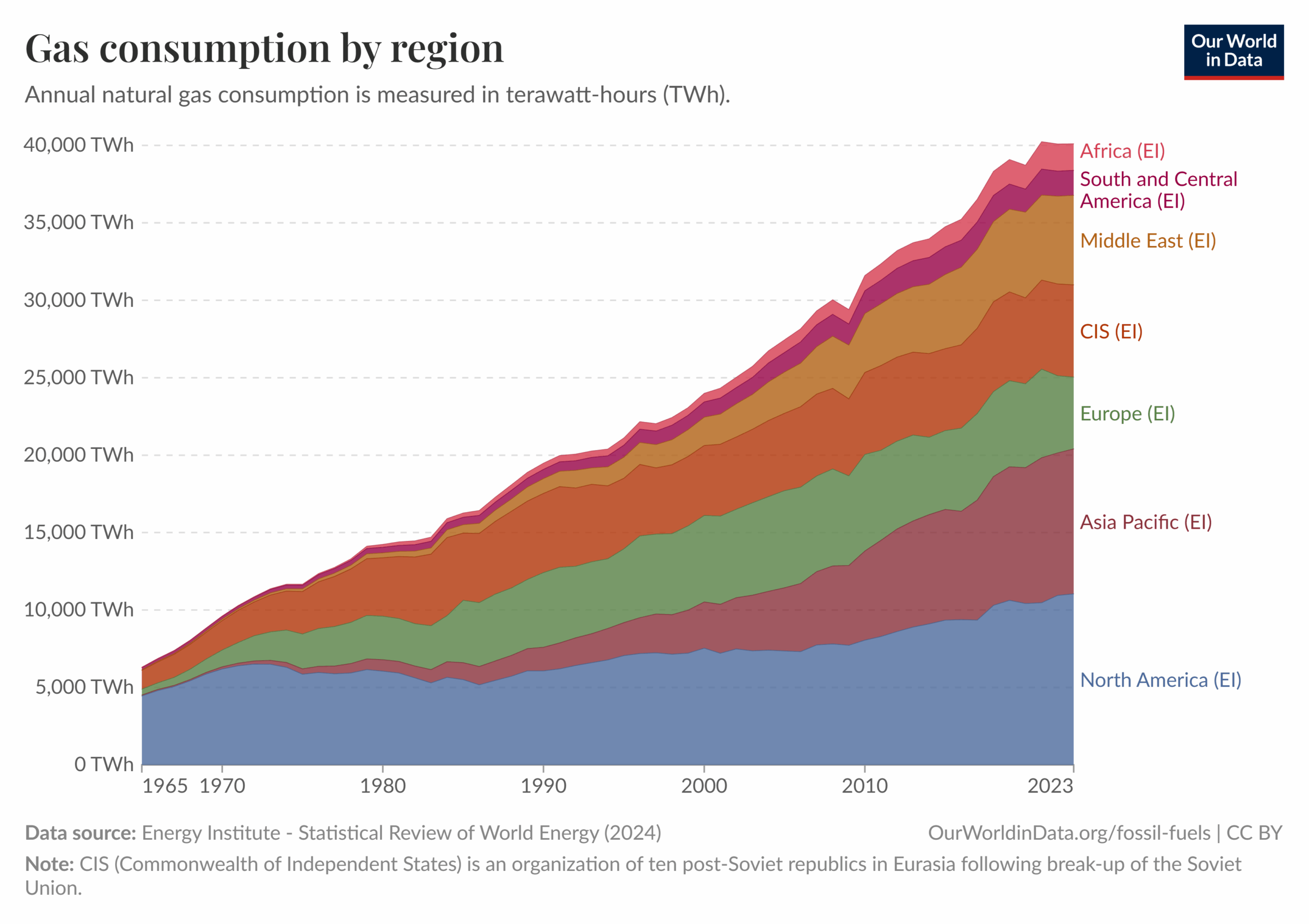
Answer Key: Question 1: (Example response: Since 1965, global natural gas consumption has steadily increased, showing that many countries rely on natural gas for their energy needs.)
Question 2: (Example response: In 1990, Europe’s natural gas consumption was much higher than Asia Pacific; but by 2020 that had changed, and Asia Pacific’s natural gas consumption overtook Europe.)
Question 3: Answers will vary. (Example: Europe continues to seek transition from fossil fuels toward renewables, as well as reduce reliance on natural gas from Russia).
Question 4: Answers will vary: (Example: Countries in Asia Pacific, such as China and India, are developing rapidly. As infrastructure investment and urbanization increase, so does the demand for and supply of natural gas as a major energy source.)
Question 5: Answers will vary.
Energy + Density Lab
Instructions: Use the Energy + Density Lab – Student Handout and the following Teacher Guide to conduct the lab activity.
Introduction
This activity is designed to help students explore and compare the concepts of physical density and energy density through hands-on investigation and visual analysis. In Part 1, students will measure mass and volume to calculate the physical density of everyday materials. In Part 2, they’ll analyze and visualize the energy density of various foods and fuels, reinforcing the idea that not all materials store or deliver energy equally. This lesson builds foundational science skills in measurement, data analysis, and critical thinking, while making connections to real-world energy use.
Student Objectives
- measure and calculate the physical density of various materials using mass and volume.
- interpret and compare the energy densities of common fuels.
- analyze and organize data to identify patterns in physical and energy density.
- make real-world connections between material properties and their practical uses.
Materials
- Student Handout (per student)
For Part 1: Introducing Physical Density (per student group of 2 or 3):
- 5 paper cups, each with a small amount of one material. Suggested materials: dried beans, glass marbles, mini metal nuts or washers, styrofoam packing peanuts and elbow or penne pasta.
- Scale (digital scale ideal) (Alternatively, to save time, teachers can pre-weigh materials and provide students with average weights for direct data entry.)
- 100-250mL graduated cylinder
- Water (for displacement)
- Calculator
For Part 2: Introducing Energy Density (per student or student group)
- 4-5 food nutrition labels
Procedure
1. Introduction: Introduce students to the definitions of physical density and energy density. These are important ideas in science that help us understand how matter and energy are distributed in space. Through a hands-on investigation and visual analysis, students will apply these concepts and see how they work in real-world examples. The table below shows the basic definitions that are also included in the Student Handout.
| Physical Density | Energy Density |
|---|---|
| Physical density (g/mL) is how much mass (g) is packed into a certain volume (mL). If something has a lot of mass in a small space, it has high physical density. If it doesn’t have much mass in a big space, it has low physical density. For example, a rock and a sponge might be the same size, but the rock feels heavy and sinks in water because it has high density. A sponge feels light and floats because it has low density. | Energy density is the amount of energy stored per unit of mass. For example, megajoules per kilogram (MJ/kg) or calories per gram (Cal/g). It tells us how much work something like fuel can give off for its size or weight. The higher the number, the more work it can do before running out. For example, a chocolate bar and a carrot might be the same size and same weight, but the chocolate bar gives you more energy because it has sugar and fat and has a higher energy density. A carrot has more water and fiber and has a lower energy density. |
2. Lab and Activities: Provide each student group the lab materials for density measurement and have them follow the instructions on the Student Handout for the physical density hands-on investigation, followed by the energy density visual analysis activities.
Sample Data and Answer Key
The Student Guide contains the Energy + Density Lab – Student questions.
Step 3
Density Data Table (Example)
| Material | Mass (g) | Initial Volume (mL) | Final Volume (mL) |
|---|---|---|---|
| Marble | 6.5 g | 80.2 mL | 82.8 mL |
| Copper Cylinder | 35.1 g | 78.5 mL | 82.5 mL |
Step 4
Calculations Density Table (Example)
| Material | Volume (mL) of material (show your work) | Density (g/mL) (show your work) |
|---|---|---|
| Marble | 82.8 mL – 80.2 mL = 2.6 mL | 6.5 g/2.6 mL = 2.5 g/mL |
| Copper cylinder | 82.5 mL – 78.5 mL = 4.0 mL | 35.1 g/4.0 mL = 8.8 g/mL |
Step 5 Questions
A. Answers will vary depending on materials used. Be certain students list all 5 materials from most dense to least dense.
B. Student answers will vary.
C. Density is used in the real world to identify materials, separate mixtures, recognize impure samples, determine buoyancy, analyze the strength of materials in engineering and construction, applied in geology for layers of earth and in atmospheric studies.
Exploring Energy Density
Step 4 Food Labels
Data Table (Example)
| Food | Energy (Cal) | Mass (g) | Energy Density (Cal/g) |
|---|---|---|---|
| Peanut butter | 190 Cal | 33 g | 5.76 Cal/g |
| 2% milk | 120 Cal | 240 g | 0.50 Cal/ g |
Step 5 Graph
Results will vary depending on the food labels examined and recorded. A bar graph would be best to display the data. Ensure that students have identified each of the 5 foods on the x-axis and used a consistent scale on the y-axis that is labeled Density (Cal/g).
Step 6 Question
High density snacks would be more compact and portable and also provide an efficient way to replenish Calories.
Step 8 Matching
- Coal
- Uranium
- Wood
- Crude Oil
- Liquified Natural Gas
STEP 9
Students should have dots equal to the energy density in each box. That is, 16 dots for wood, 24 dots for coal, 42 dots for crude oil, 55 dots for liquified natural gas, and the complete box colored in for uranium.
Step 10 Reflection Questions
- Student answers will vary.
- Energy density of food is in Calories per gram while energy density of fuels is in MJ/kg, although both represent energy (calories and Joules both measure energy) per unit of mass.
- A fuel with low energy density is wood while a food with low energy density is milk (food answer will vary depending on food samples used).
- Of the fuels listed in the question, uranium (nuclear) is used the least globally despite having a very high energy density.
- Oil is used the most globally. Of the non-nuclear fuels, it has a fairly high energy density.
- Eating foods high in energy density make you feel full for a longer period of time. Batteries with high energy density may be smaller and more portable. Crude oil is made into gasoline and other transportation fuels and can supply a large amount of energy.
- Liquified natural gas has a higher energy density than coal and oil. It can then provide more energy per mass than coal and oil. Other advantages include fewer emissions of carbon dioxide and particulate matter. Natural gas also does not create ash like coal.
- Natural gas is a good choice for heating and cooking in homes because of its high energy density, meaning it releases a lot of heat per unit volume when burned. It would be efficient in heating large spaces. It is clean burning with less production of carbon dioxide, nitrogen oxides and no sulfur oxides.
- Student answers will vary but may reflect the high energy density of natural gas as well as the versatility of natural gas. Students may express not to use natural gas as it still produces carbon dioxide emissions.
Exit Ticket
Instructions: Access the Exit Ticket and have students reflect on and answer the prompt.
