Part 1: Introducing Physical Density
Physical density (g/mL) is how much mass (g) is packed into a certain volume (mL). If something has a lot of mass in a small space, it has high physical density. If it doesn’t have much mass in a big space, it has low physical density.
Measuring Physical Density
Step 1: Gather the materials that your teacher provided for you. List each material that you will be measuring the physical density of in the first column of both data tables.
Step 2: If your teacher is providing you with the mass (grams) of each object, record it in the first data table. If you measure the mass yourself, follow the instructions below.
- Turn on the scale, add a piece of a material, and measure its mass in grams. Record the mass of each material on the first data table.
Step 3: Next, you will measure the volume of the material using a graduated cylinder and water.
- Fill a graduated cylinder with approximately 80 mL of water but write down the EXACT amount in the Initial Volume column of the Density Data Table.
- Gently add the material to the graduated cylinder, submerging it in the water.
- Record the new volume in the Final Volume column of the Density Data Table.
- Subtract the final volume from the initial volume to determine the volume of the material. Show your work in the Calculations Density Table.
Density Data Table
| Material | Mass (g) | Initial Volume (mL) | Final Volume (mL) |
|---|---|---|---|
Step 4: Now you are ready to calculate the physical density of each material! Use the formula:
Density (g/mL) = Mass (g) / Volume (mL). Show your work for each material in the Calculations Table.
Calculations Density Table
| Material | Volume (mL) of material (show your work) | Density (g/mL) (show your work) |
|---|---|---|
Step 5: Answer the questions.
- List each material and their physical densities, from highest to lowest.
- Did any of the results surprise you? Why?
- How do you think measuring physical density helps us in the real world? (e.g. in construction, shipping, environmental science, etc.)
Part 2: Introducing Energy Density
Energy density is the amount of energy stored in a fuel per unit of mass. It tells us how much work you can get out of a certain amount of fuel or battery. For example, a chocolate bar and a banana might be the same size, but the chocolate bar gives you more energy – so it has a higher energy density.
To calculate the energy density of biomass and fossil fuels, we find the megajoules (energy) per kilogram (unit of mass). To calculate the energy density of food, we find the calories (energy) per gram (unit of mass). Food is fuel for our bodies, and we consume foods with different energy densities every day. Your body “burns” food the same way a car burns fuel – it just does it chemically and more slowly. In this activity, you will use nutrition labels to calculate the energy density of food.
Exploring Energy Density
Step 1: Gather the nutrition labels and list the names from each label in the Food column of the table.
Step 2: Carefully look at the calories of one serving size on the labels. Record the calories per serving size in the Energy column.
Step 3: Carefully find the grams per serving size on the labels. Record it in the Mass column.
Step 4: Now you are ready to calculate the energy density of your foods! Use the formula:
Energy Density (Cal/g) = Energy (Cal) / Mass (g). Record your answers for each food in the Energy Density column.
Data Table
| Food | Energy (Cal) | Mass (g) | Energy Density (Cal/g) |
|---|---|---|---|
Step 5: Plot a graph or create a ranking of the foods, from highest to lowest energy density.

Step 6: Answer the question.
- Why do athletes or hikers often choose high energy-density snacks?
Step 7: Now, let’s turn our attention to the energy density of various fuels. Just like the calories of food vary, the energy of fuels also vary. We measure the energy density of fuels with megajoules per kilogram. Study the following data table, comparing the approximate energy density of various fuels.
| Fuel | Energy Density (MJ/kg) |
|---|---|
| Wood | ~15 MJ/kg |
| Coal (Bituminous) | ~24 MJ/kg |
| Crude Oil | ~42 MJ/kg |
| Liquified Natural Gas | ~55 MJ/kg |
| Uranium (enriched to 3.5%, in a Light Water Reactor) | ~3,900,000 MJ/kg |
Step 8: Match the fuel with the description of its energy density. Use each fuel type only once.
- _________________________ More energy than wood. Common in power plants.
- _________________________ Extremely high! A tiny amount gives huge energy. Used in nuclear.
- _________________________ Low energy for its weight. You need a lot to make power.
- _________________________ High energy. Used for gasoline and fuel.
- _________________________ High energy, especially in liquid form.
Step 9: Imagine that each container represents the same volume of a certain fuel. With your pencil or pen, add one dot to represent EACH MJ/kg of energy density for the first four fuels. When you get to uranium, just color in the whole box!

Step 10: Answer the reflection questions.
A. Did any of the energy densities of the foods and fuels surprise you? Why?
B. Compare how to calculate energy density of foods to the energy density of fuels.
C. Can you identify a fuel and a food that both have low energy density?
D. Study the graph below. Out of wood (traditional biomass), coal, oil, natural gas and uranium (nuclear), which fuel’s energy is consumed the least globally? Does it have high or low energy density?
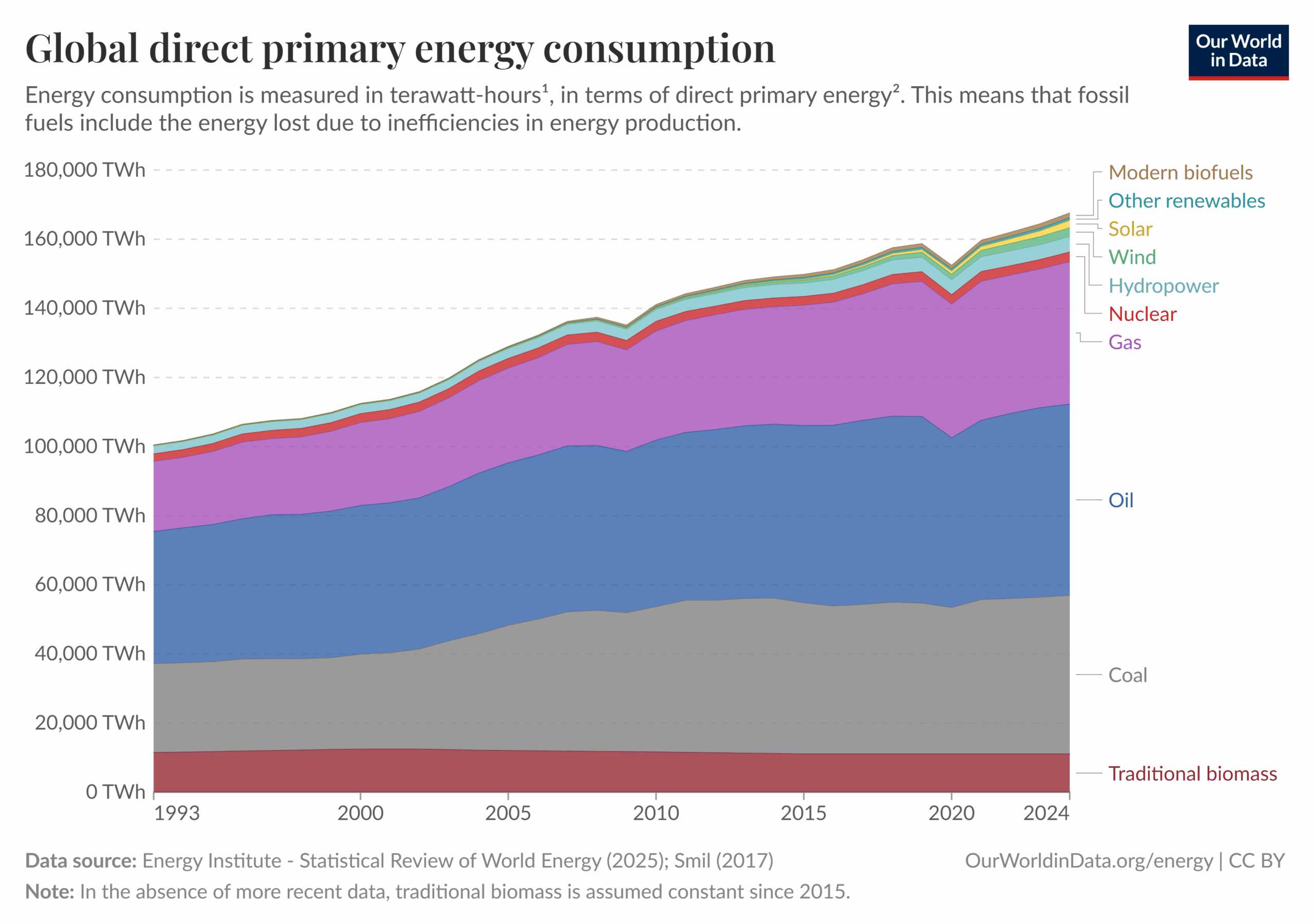
E. Using the same graph, identify which fuel’s energy is consumed the most globally. Does it have high or low energy density?
F. Can you think of examples where energy density is important in your everyday life?
Natural Gas Focus
G. How does the energy density of natural gas compare to other fuels like coal and oil? What are some advantages of using natural gas over coal and wood as a fuel?
H. Natural gas is often used for heating homes and cooking. How does its energy density make it a good choice for these uses?
I. If you were designing an energy system for a city, would you include natural gas? Why or why not?
